The total energy of the system, which contains the vacancy cluster not filled by helium E0 was calculated by averaging over the simulation time. The total energy of the system, which contains the vacancy cluster, filled with a necessary quantity of atoms of helium E was calculated in the same way. Dissolution energy is equal to a difference in the total potential energies for the system with the filled and not filled by helium vacancy cluster at the zero absolute temperature.
![]() . (4)
. (4)
3. Results of the simulation of helium dissolution in Pd vacancy clusters and their discussion
As a result of the simulation of the dissolution of helium in the vacancy clusters of palladium, which consist of a different quantity of vacancies (8÷10) and which have the different configurations (see Fig. 4), energy of the dissolution ![]() were obtained, where (j) is number of the helium atoms in vacancy clusters and, correspondingly, is equal (j) = 1, 2, 3 and 4. For the comparison with the experimental data the values of dissolution energies for the dissolution of each j-th helium atom newly added to the vacancy cluster already containing (j - 1) dissolved helium atoms, were calculated.
were obtained, where (j) is number of the helium atoms in vacancy clusters and, correspondingly, is equal (j) = 1, 2, 3 and 4. For the comparison with the experimental data the values of dissolution energies for the dissolution of each j-th helium atom newly added to the vacancy cluster already containing (j - 1) dissolved helium atoms, were calculated.
![]() . (6)
. (6)
Results of calculating dissolution energies and their comparison with the experimental data are shown at table 3. The calculation procedure based on the example of the dissolution (1¸4) helium atoms in the vacancy cluster, which consists of 9 vacancies, is illustrated at table 4.
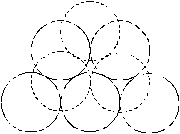
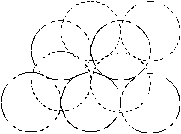
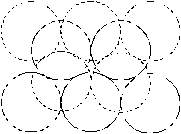
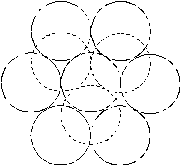
The simulation of the helium dissolution in vacancy clusters with the size of 8¸10 vacancies (potential IV) reproduces well both the dissolution energies of the first two helium atoms and change of the sign of dissolution energy for third and the fourth helium atoms. For the cluster of 8 vacancies the value of the dissolution energy for fourth atom of helium is clearly overstated for both used potentials, whereas helium dissolution in the cluster of 9 vacancies gives a good agreement of the dissolution energy value for the third and fourth atom with the experimental data (potential IV). Apparently, configuration and size of real grain-boundary vacancy clusters are close to the model cluster of 9 vacancies (Fig. 4, c). A difference for the different potentials He-Pd (II and IV) of dissolution energies for second - fourth atoms in the identical clusters, is resulted from different depth and extent of the well of these potentials (fig. 3).
|
|
a) | b) |
|
|
c) | d) |
Fig. 4. Configurations of the vacancy clusters, shown in the [111] direction. Solid line denotes the particles, which lie at the plane of paper; broken lines denote particles above the plane of paper in the adjacent hexagonal planes. a) - 8 vacancies; b) - 9 vacancies; c-dvacancies. |
Table 3.
Calculation and comparison with the experiment of dissolution energy of the helium atoms in the clusters formed by (8÷10) vacancies with the use of potentials II and IV
j |
| Dissolution energy | ||||||
8 vacancies | 9 vacancies | 10 vacancies | ||||||
II | IV | II | IV | II* | IV* | IV** | ||
1 | -0.028 ± 0.005 | -0.022 | -0.022 | -0.024 | -0.026 | -0.023 | -0.025 | -0.024 |
2 | -0.010 ± 0.003 | -0.024 | -0.005 | -0.023 | -0.013 | -0.023 | -0.020 | -0.003 |
3 | +0.050 ± 0.006 | -0.006 | +0.014 | -0.008 | +0.014 | -0.009 | +0.011 | -0.011 |
4 | +0.070 ± 0.007 | +0.129 | +0.155 | +0.026 | +0.052 | -0.009 | +0.015 | +0.034 |
Configurations of clusters consisting of 10 vacancies are given: * - at Fig. 4, c; ** - at Fig. 4, d.
Table 4.
Calculation and comparison of dissolution energy of helium atoms in the vacancy cluster (9 vacancies) with the experimental values (potential IV)
The number of helium atoms in the cluster j |
|
|
|
|
0 | -5487.15976 | - | - | - |
1 | -5487.18569 | -0.02593 | -0.02593 | -0.028 ± 0.005 |
2 | -5487.19869 | -0.03893 | -0.01300 | -0.010 ± 0.003 |
3 | -5487.18421 | -0.02445 | +0.01448 | +0.050 ± 0.006 |
4 | -5487.13213 | +0.02763 | +0.05208 | +0.070 ± 0.007 |
Here E is the total energy of the system, which contains cluster where dissolves helium atoms, ![]() are experimental values of energy of dissolution (Table 1). The error of total energy and other values determination influences the last significant place only.
are experimental values of energy of dissolution (Table 1). The error of total energy and other values determination influences the last significant place only.
The change of sign ![]() with the temperature for the first and second stages (table 1) can be resulted from either a change of configuration of the vacancy cluster, in which the dissolution occurs, with the increase of saturation temperature (for example, the detachment of vacancies from the cluster), or the presence of other dissolution positions with the increased dissolution energy in the limits of the cluster, which are filled by helium only at a higher temperature of saturation.
with the temperature for the first and second stages (table 1) can be resulted from either a change of configuration of the vacancy cluster, in which the dissolution occurs, with the increase of saturation temperature (for example, the detachment of vacancies from the cluster), or the presence of other dissolution positions with the increased dissolution energy in the limits of the cluster, which are filled by helium only at a higher temperature of saturation.
Obtained computed values of dissolution energy are in the satisfactory agreement with the experimental values. This indicates the applicability of MD method in the solution of similar problems.
It is shown that the minimum value of experimental energy of dissolution is determined by interaction of the helium atom with the surface of free cluster. The registered in MD experiment change of the helium dissolution energy with an increase of the number of helium atoms in the cluster is in accordance with the proposed dissolution model.
On the basis of the proposed model the concentration of vacancy clusters was determined, which for this sample is equal 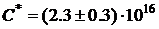 cm-3.
cm-3.
The authors thank prof. Mulyukov R. R. for the palladium samples supplied for the study, and Potashnikov S. I. for the calculation of He-Pd potential by the density functional technique.
References
1. Zhiganov A. N., Kupryazhkin A. Ya., Mulyukov R. R., Bitkulov I. Kh. // ZhTF 1998. V. 72. № 11. P. 96‑99.
2. Nazarov A. A. and Mulyukov R. R. Nanostructured Materials. In: Handbook of Nanoscience, Engineering, and Technology, Ed. Goddard W., Brenner D., Lyshevsk S., Iafrate G., CRC Press. 2002.
3. Mulyukov R. R., Starostenkov M. D. // Acta Met. Sinica. 2000. V. 13. № 1. P. 301‑309.
4. Smirnova N. A., Levit V. I., Pilyugin V. P., etc. // FMM. 1986. V. 61. P. 1170.
5. Zolotarevskiy V. S. M.: Metallurgy. 19 P.
6. Würschum R., Kübler A. et. al. // Ann. de Chim. - Sci. des Mat. 1996. V. 21. P. 471‑482.
7. Kupryazhkin A. Ya., Kurkin A. Yu., Kitayev G. A. // ZhFKh. 1988. Т. 62. № 12. С. .
8. Wilson W. D., and Johnson R. A. “Rare gas in metals”, In Interatomic potentials and simulation of lattice defects. edited by P. C. Gehlen et. al., Plenum Press, New York – London, 1972, P. 375‑385.
9. Mezhfazovaya granitsa gas – tverdoe telo. Edited by E. Flood. M.: Mir. 19 P.
10. Varaksin A. N., Kozyaychev V. S. // FMM. 1991. № 2. P. 45-51.
11. Shu Zhen, Davies G. J. // Phys. stat. sol. (a). 1983. V. 78. № 72. P. 595‑605.
12. Hirschfelder J., Curtiss Ch., Bird R. Molekulyarnaya teoriya gazov i zhidkostey. M.: Izdatel'stvo inostrannoy literatury. 19 P.
13. Michels, A., Wouters, H. // Physica. 1941. V. 8. P. 923
14. Delly B. // J. Chem. Phys. 1986. V. 92. P. 329.
15. Kim Y. S., Gordon R. G. // J. Chem. Phys. 1974. V. 61. № 1, P. 1-16.
16. Smith K. M., Rulis A. M., Scoles G. et. al. // J. Chem. Phys. 1977. V. 67. № 1, P. 152-163.
17. Zaremba E., Kohn W. // Physical rewiev B. 1977. V. 15. № 4, P. .

|
Из за большого объема этот материал размещен на нескольких страницах:
1 2 3 |